Chlorzoxazone USP
Chlorzoxazone USP Specification
- Molecular Formula
- C7H4ClNO2
- HS Code
- 2924 29 90
- Place of Origin
- India
- Loss on Drying
- 0.5% max
- EINECS No
- 202-411-2
- Assay
- 99.0% to 101.0%
- Heavy Metal (%)
- <0.001%
- Residue on Ignition
- 0.1% max
- Melting Point
- 189C to 193C
- Moisture (%)
- 0.5% max
- Particle Size
- Passed through 60 mesh
- Molecular Weight
- 169.57 g/mol
- Storage
- Store in a cool, dry place, protected from light
- Other Names
- Chlorzoxazone, 5-Chloro-2-benzoxazolinone
- CAS No
- 95-25-0
- Type
- Pharmaceutical Raw Material
- Grade
- USP
- Usage
- Muscle Relaxant
- Purity
- 99% min
- Appearance
- White to off-white crystalline powder
- Application
- Active Pharmaceutical Ingredient
- Raw Material
- Yes
- Smell
- Odorless
- Color
- White
- Form
- Powder
- Total impurities
- 0.3%
- Packaging
- Packed in HDPE drums, double polyethylene bags inside
- Shelf Life
- 36 months from date of manufacture
- Certificate of Analysis
- Available upon request
- Microbial Limit
- Meets USP microbial requirements
- Solubility
- Slightly soluble in water, freely soluble in alcohol and in chloroform
- Identification
- Meets USP requirements (IR, TLC) for Chlorzoxazone
- Maximum single impurity
- 0.1%
Chlorzoxazone USP Trade Information
- Minimum Order Quantity
- 500 Kilograms, ,
- Supply Ability
- 10000 Kilograms, , Per Month
- Delivery Time
- 15 Days
- Main Domestic Market
- All India
About Chlorzoxazone USP
Exceptional Quality and Compliance
Chlorzoxazone USP is manufactured under stringent quality controls, adhering to all USP standards, including limits for impurities and microbial content. Each shipment is thoroughly tested for identity by IR and TLC, ensuring only pure and potent material reaches you.
Optimal Packaging for Extended Shelf Life
To ensure utmost protection, Chlorzoxazone USP is sealed in HDPE drums lined with double polyethylene bags. This packaging helps maintain product quality during transport and storage, providing a shelf life of up to 36 months from manufacturing date.
Versatile Solubility and Application
Slightly soluble in water but freely soluble in alcohol and chloroform, Chlorzoxazone USP is highly adaptable for various pharmaceutical formulations. Its use as an active pharmaceutical ingredient in muscle relaxant medications highlights its importance in medical manufacturing.
FAQ's of Chlorzoxazone USP:
Q: How should Chlorzoxazone USP be stored to maintain quality?
A: Chlorzoxazone USP should be stored in a cool, dry place, protected from light, and kept in its original packaging to maintain its purity and efficacy throughout its 36-month shelf life.Q: What is the primary usage of Chlorzoxazone USP?
A: Chlorzoxazone USP is primarily used as an active pharmaceutical ingredient in muscle relaxant medications, helping to relieve muscle spasms and discomfort.Q: When is a Certificate of Analysis provided for this product?
A: A Certificate of Analysis is available upon request with each batch, ensuring transparency and verifying compliance with all USP and quality parameters.Q: Where is Chlorzoxazone USP manufactured and exported from?
A: This product is manufactured and supplied from India, with availability for export, trading, and wholesale supply to pharmaceutical companies worldwide.Q: What processes ensure the purity and safety of Chlorzoxazone USP?
A: Each batch undergoes comprehensive analysis, including IR and TLC identification, impurity testing, and microbial limit assessments as per USP standards, ensuring high purity and safety.Q: What are the main benefits of using Chlorzoxazone USP in pharmaceutical applications?
A: Chlorzoxazone USP offers high purity, excellent solubility in common solvents, and reliable compliance with international pharmacopoeia requirements, making it ideal for consistent pharmaceutical formulations.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Drugs Intermediates Category
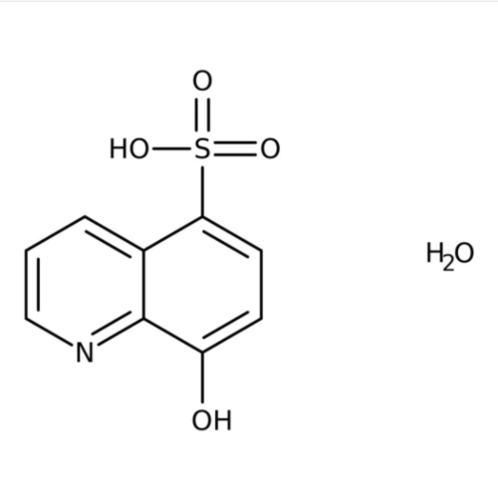
8-hydroxy quinoline 5-sulfonic acid monohydrate
Price 8000-9000 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Color : Other, Pale yellow to beige
Storage : Other, Store in cool, dry, wellventilated area in tightly closed containers
Form : Other, Crystalline powder
Purity : 98%
Myristyl dimethyl benzyl ammonium chloride powder
Price 2500 INR / Kilograms
Minimum Order Quantity : 500 Kilograms
Color : White
Storage : Other, Store in a cool, dry, wellventilated area away from strong oxidizers
Form : Powder
Purity : >98%
3 Nitrophthalonitrile
Price 3250-3350 INR / Kilograms
Minimum Order Quantity : 100
Color : Other, Light yellow to pale brown
Storage : Other, Keep tightly sealed in a cool, dry place
Form : Solid
Purity : 99%
Halquinol 98%
Price 1000 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Color : White
Storage : Room Temperature
Form : Powder
Purity : 98%